JEE Advanced | 2013Solid StateHard
Question
The arrangement of X- ions around A+ ion in solid AX is given in the figure (not drawn to scale).If the radius of X- is 250 pm, the radius of A+ is -
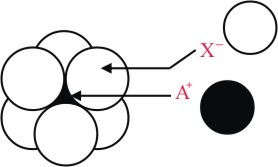

Options
A.104 pm
B.125 pm
C.183 pm
D.57 pm
Solution
Limiting radius ratio for octahedral void is 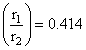
r+ = 0.414 × 250 pm ≃ 104 pm
Correct option (A)
r+ = 0.414 × 250 pm ≃ 104 pm
Correct option (A)
Create a free account to view solution
View Solution FreeMore Solid State Questions
Aluminum metal has a density of 2.72 g/cm3 and crystallizes in a lattice with an edge length of 404 pm.Which of the foll...In a face centerd lattice of X and Y, X atoms are present at the corners while Y atoms are at face centers. Then the for...If the height of HCP unit cell of identical particles is h, then the height of octahedral voids from the base is...Frenkel defect is observed in...There are three cubic unit cells A, B and C. A is FCC and all of its tetrahedral voids are also occupied. B is also FCC ...