Periodic TableHard
Question
The first ionisation potential of Na is 5.1 eV. The value of electron gain enthalpy of Na+ will be :-
Options
A.- 2.55 eV
B.- 5.1 eV
C.- 10.2 eV
D.+ 2.55 eV
Solution
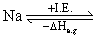 Na+ + e-
Na+ + e- |I.E.| = |ᐃHe.g.| Hence
ᐃHe.g. of Na+ = - 5.1 eV
Create a free account to view solution
View Solution FreeMore Periodic Table Questions
The electrongativity of the following elements increases in the order :...Among Al2O3, SiO2, P2O3 and SO2 the correct order of acid strength is...The set representing the correct order of first ionisation potential is :...Which of the following shows false order :-...The formation of the oxide ion O2-(g) requires first an exothermic and then an endothermic step as shown below O(g) + e-...