Periodic TableHard
Question
The electrongativity of the following elements increases in the order :
Options
A.C, N, Si, P
B.N, Si, C, P
C.Si, P, C, N
D.P, Si, N, C
Solution
On moving L → R across a period electronegatively increases, whil on moving down in a group it decreases. In the periodic table relative position of these element is
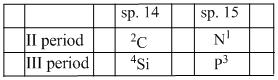
thus correct order is Si < P < C < N

thus correct order is Si < P < C < N
Create a free account to view solution
View Solution FreeMore Periodic Table Questions
In the long form of the periodic table, the valence shell electronic configuration of 5s25p4 corresponds to the element ...Which one the following sets of ions represents the collection of isoelectronic species?...In which of the following process energy is absorbed:...Which of the following does not have hemiacetal group :-...An element has electronic configuration [Xe] 4f7 5d1 6s2. It belong to the.....block of the periodic table....