Redox and Equivalent ConceptHard
Question
Consider the following reaction:
x MnO4- + y C2O42- + z H+ → x Mn2+ + (2y) CO2 + (z/2) H2O
The values of x, y and z in the reaction are respectively :-
Options
A.5,2 and 16
B.2,5 and 8
C.2, 5 and 16
D.5,2 and 8
Solution
(i) MnO4- + 8H+ + 5e- → Mn+2 + 4H2O
(ii) C2O4-2 → 2CO2 + 2e-
⇒ (i)2 + (ii) 5

2MnO4- + 16H+ + 5C2O4-2 → 2Mn+2 + 8H2O + 10CO2

Given equation :
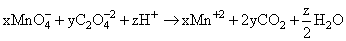
On comparing with balanced equation
we get ; x = 2, y = 5, z = 16.
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
A mole of N2H4 loses 10 mol of electrons to from a new compound y. Assuming that all the nitrogen appears in the new com...Identify the correct statements :A. Hydrated salts can be used as primary standard.B. Primary standard should not underg...A quantity of 0.10 g of anhydrous organic acid requires 25 ml of 0.10 N-NaOH for neutralization. A quantity of 0.245 g o...The heat required to raise the temperature of body by 1 K is called...Which of the following ion cannot act as an oxidizing agent?...