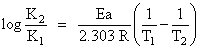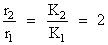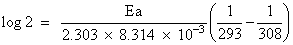Chemical Kinetics and Nuclear ChemistryHard
Question
What is the activation energy for a reaction if its rate doubles when the temperature is raised from 20oC to 35oC ? (R = 8.314 J mol-1 K-1)
Options
A.15.1 kJ mol-1
B.342 kJ mol-1
C.269 kJ mol-1
D.34.7 kJ mol-1
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Which of the following ions will have the minimum coagulating value for the sol obtained by adding FeCl3 solution to sli...3A → B + CIt would be a zero order reaction when :...At a certain temperature, the first order rate constant k1 is found to be smaller than the second order rate constant k2...Under the same reaction conditions, initial concentration of 1.386 mol dm-3 of a substance becomes half in 40 seconds an...In compounds of type ECl3 where E = B, P. As or Bi, the angles Cl − E − Cl for different R in order-...