Mole ConceptHard
Question
Vapourisation of CCl4 occurs reversibly at 27oC and 1 atm
CCl4(l) → CCl4(g) ; ᐃHvap = 42.0 kJ/mole at 27oC and 1 atm
If 1 mole of liquid CCl4 at 27oC has entropy of 214J/K-mole, what is the entropy (in J/K-mol) of 1 mole of vapour in equilibrium with liquid at this temperature.
CCl4(l) → CCl4(g) ; ᐃHvap = 42.0 kJ/mole at 27oC and 1 atm
If 1 mole of liquid CCl4 at 27oC has entropy of 214J/K-mole, what is the entropy (in J/K-mol) of 1 mole of vapour in equilibrium with liquid at this temperature.
Options
A.74
B.454
C.354
D.254
Solution
If liquid CCl4 is in equilibrium with it′s vapour′s them
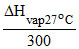 = ᐃSvap = SCCl4(g) - SCCl4(l)
= ᐃSvap = SCCl4(g) - SCCl4(l)
⇒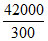 = SCCl4(g) - 214J/Kmole
= SCCl4(g) - 214J/Kmole
140 + 214 = 354 J/K mole.
⇒
140 + 214 = 354 J/K mole.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
25 mL of an aqueous solution of KCl was found to require 20 mL of 1 M AgNO3 solution when titrated using a K2CrO4 as ind...Total charge required for the oxidation of two moles Mn3O4 into MnO4-2 in presence of alkaline medium is :-...The vapour density of gas A is four times that of B. If molecular mass of B is M, then the molecular mass of A is :-...Which of the following is/are examples of uni molecular reactions ?...In a binary ideal liquid solution of A and B in which vapour pressure of pure component A and B are 600 and 400 torr res...