Wave motionHard
Question
Which one of the following pairs is isostructural (i.e., having the same shape and hyridization) ?
Options
A.[ BCl3 and BrCl3 ]
B.[ NH3 and NO3- ]
C.[ NF3 and BF3 ]
D.[ NF4- and BF4- ]
Solution
If the number of bond pairs and lone pairs are same for the given pairs, they are isostructural.
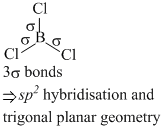
(a) BCl3 and BrCl3
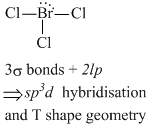
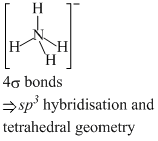
(b) NH3 and NO3-

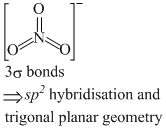
(c) NF3 and BF3

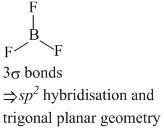
(d) BF4- and NH4-
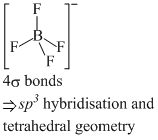
(a) BCl3 and BrCl3

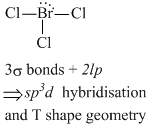
(b) NH3 and NO3-


(c) NF3 and BF3


(d) BF4- and NH4-


Create a free account to view solution
View Solution FreeMore Wave motion Questions
For given equation y = 10-3 sin (4000 πt + x) determine time period....The wavelength of the light received from a galaxy is 0.4% greater than the wave length on the earth then the velocity o...Following are equations of four waves :(i) 1 = a sin ω (ii) y2 = a cos ω(iii) z1 = a sin ω (iv) z2 = a co...In a resonance tube the first resonance with a tuning fork occurs at 16 cm and second at 49 cm. If the velocity of sound...Doppler effect for light differs from that for sound in regards that :...