Math miscellaneousHard
Question
The enthalpy of fusion of water is 1.435 kcal/mol. The molar entropy change for the melting of ice at 0oC is
Options
A.10.52 cal/(mol K)
B.21.04 cal/(mol K)
C.5.260 cal/(mol K)
D.0.526 cal/(mol K)
Solution
Molar entropy change for the melting of ice,
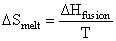
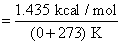
= 5.26 × 10-3 kcal / mol K
= 5.26 cal / mol K
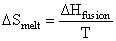
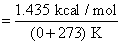
= 5.26 × 10-3 kcal / mol K
= 5.26 cal / mol K
Create a free account to view solution
View Solution FreeMore Math miscellaneous Questions
Number of divisors of 1024 × (225)2 × (25)7 which are of the form 3k + 1 (where k is a whole number) is equal ...Evaluate dx,x > 0...If one root of the equation ax2 + bx + c = 0 is square of the other, then the value of b3 + ac2 + a2c is :...Let |a| 2 - |α|x-|β|=0. If|α| |a| - 1 = 0...If is the adjoint of a 3 × 3 matrix A and |A| = 4, then α is equal to...