SolutionHard
Question
The resistance of a solution ′A′ is 40 ohm and that of solution ′B′ is 70 ohm, both solutions being taken in the same conductivity cell. If equal volumes of solutions ′A′ and ′B′ are mixed, what will be the resistance of the mixture using the same cell ? (Assume that there is no increase in the degree of dissociation of ′A′ and ′B′ on mixing)
Options
A.50.9 ohm
B.101.8 ohm
C.110 ohm
D.55 ohm
Solution
For solu. ′A′ : K = 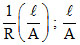 = x(cell const.)
= x(cell const.)
K1 =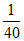 × x
× x
For solu. ′B′ : K2 =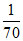 × x
× x
If equal vol. of solu. are mixed then vol. becomes doubled & conductivity of mix. becomes halved.
∴ Conductivity of the mix. will be
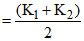
∴ for the mix. :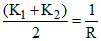 × x
× x
R → Resistance of the mix.
K1 =
For solu. ′B′ : K2 =
If equal vol. of solu. are mixed then vol. becomes doubled & conductivity of mix. becomes halved.
∴ Conductivity of the mix. will be
∴ for the mix. :
R → Resistance of the mix.
Create a free account to view solution
View Solution FreeMore Solution Questions
The osmolarity of 0.2 M-Na2SO4 is...The colligative properties of a solution may depend on...The depression of freezing points of 0.05 molal aqueous solution of the following compounds are measured.1. NaCl 2. K2SO...Consider following cases -I : 2M CH3COOH solution in benzene at 27oC where there is dimer formation to the extent of 100...An azeotropic solution of two liquids has boiling point lower than either of them when it :...