Heat and Thermal ExpansionHard
Question
The molar specific heat of the the process V ∝ T4 for CH4 at room temperature is : -
Options
A.4 R
B.7 R
C.3 R
D.8 R
Solution
V ∝ T4 ⇒ V ∝ (PV)4
⇒ P4V3 = contant ⇒ PV3/4 = constant
∴ C = Cv +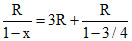 =3R + 4R = 7R
=3R + 4R = 7R
⇒ P4V3 = contant ⇒ PV3/4 = constant
∴ C = Cv +
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A cup of tea cools from 80oC to 60oC in one minute.the ambient temperature is 30oC. In cooling from 60oC to 50oC. will t...A steel tape is placed around the earth at the equator when the temperature is 10oC. What will be the clearance between ...A sample of gas follows process represented by PV2 = constant. Bulk modulus for this process is B, then which the of the...If S is stress and Y is Young′s modulus of material of a wire, the energy stored in the wire per unit volume is...N(< 100) Molecules of a gas have velocities 1, 2, 3.........N , km/s respectively . Then :-...