Solid StateHard
Question
In a solid ′AB′ having the NaCl structure ′A′ atom occupy the corners of the cubic unit cell. If all the face centre atoms along one of the axes are removed then the resultant stoichiometry of the solid is :
Options
A.AB2
B.A2B
C.A4B3
D.A3B4
Solution
A ⇒ 8 × 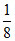 + 4 ×
+ 4 × 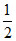 × = 3
× = 3
B ⇒ 12 ×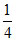 + 1 = 4
+ 1 = 4
A3B4
B ⇒ 12 ×
A3B4
Create a free account to view solution
View Solution FreeMore Solid State Questions
The composition of a sample of cuprous oxide is found to be Cu1.92O1.00 due to metal deficient defect. The molar ratio o...Lithium borohydride (LiBH4) crystallizes in an orthorhombic system with four molecules per unit cell. The unit cell dime...A metal crystallizes in such a lattice in which only 70% of the total space of the crystal is occupied by the atoms. If ...Correct statement for ccp is :...A metal crystallizes with a face-centered cubic lattice. The edge of the unit cell is 408 pm. The diameter of the metal ...