CalorimetryHard
Question
An ideal gas with pressure P, volume V and temperature T is expanded isothermally to a volume 2V and a final pressure PI. The same gas is expanded adiabatically to a volume 2V, the final pressure is PA. In terms of the ratio of the two specific heats for the gas γ, the ratio PI/PA is:
Options
A.2γ-1
B.21-γ
C.2γ
D.2γ
Solution
As gas expands isothermelly
so P1 =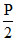
In adiabatic process PVγ = constant
so PVγ = PA(2V)γ
⇒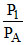 = 2γ-1
= 2γ-1
so P1 =
In adiabatic process PVγ = constant
so PVγ = PA(2V)γ
⇒
Create a free account to view solution
View Solution FreeMore Calorimetry Questions
If I is the moment of inertia of a solid body having a -coefficient of linear expansion then the change in I correspondi...If a bimetallic strip is heated, it will...A small quantity, mass m, of water at a temperature q (inoC) is poured on to a large mass M of ice which is at its melti...A point mass is subjected to two simultaneous sinusoidal displacements in x-direction, x1(t) = A sin ωt and x2(t) =...A metal ball immersed in water weighs w1 at 5oC and w2 at 50oC. The coefficient of cubical expansion of metal is less th...