Basic Maths and Units and DimensionsHard
Question
An ideal gas is expanding such that PT2 = constant. The coefficient of volume expansion of the gas is:
Options
A.1/T
B.2/T
C.3/T
D.4/T
Solution
PT2 = constant and PV = nRT
implies V ∝ T3
⇒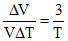
⇒ γ =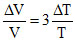
implies V ∝ T3
⇒
⇒ γ =
Create a free account to view solution
View Solution FreeTopic: Basic Maths and Units and Dimensions·Practice all Basic Maths and Units and Dimensions questions
More Basic Maths and Units and Dimensions Questions
Two samples A and B of a gas initially at the same pressure and temperature are compressed from volume V to V/2 (A isoth...An air column, closed at one end and open at the other, resonates with a tuning fork when the smallestlength of the colu...Two physical quantities of which one is a vector and the other is a scalar having the same dimensional formula are :...A ring is cut from a platinum tube 8.5 cm internal and 8.7 cm external diameter. It is supported horizontally from the p...What should be the maximum acceptance angle at the air-core interface of an optical fibre if n1 and n2 are the refractiv...