Basic Maths and Units and DimensionsHard
Question
Two samples A and B of a gas initially at the same pressure and temperature are compressed from volume V to V/2 (A isothermally and adiabatically). The final pressure of A is :-
Options
A.Greater than the final pressure of B
B.Equal to the final pressure of B
C.Less than the final pressure of B
D.Twice the final pressure of B
Solution
A is compressed isothermally, hence
P1V = P2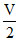 ⇒ P2 = 2P1
⇒ P2 = 2P1
and B is compressed adiabatically, hence
P1′ Vγ = P′2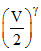 ⇒ P′2 = (2)γ P′1
⇒ P′2 = (2)γ P′1
Since γ > 1, hence P2′ > P2 or P2 < P2′
P1V = P2
and B is compressed adiabatically, hence
P1′ Vγ = P′2
Since γ > 1, hence P2′ > P2 or P2 < P2′
Create a free account to view solution
View Solution FreeTopic: Basic Maths and Units and Dimensions·Practice all Basic Maths and Units and Dimensions questions
More Basic Maths and Units and Dimensions Questions
A constant voltage is applied between the two ends of a uniform metallic wire. Some heat is developed in it. If both len...Two skaters A and B of mass 50 kg and 70 kg, respectively, stand facing each other, 6 metres apart on a horizontal smoot...All the rods have same conductance ′K′ and same area of cross section ′A′. If ends A and C are m...A point source S is placed at the bottom of a transparent block of height 10 mm and refractive index 2.72. It is immerse...An arrangement of three parallel straight wires placed perpendicular to plane of paper carrying same current 'I alon...