Chemical Kinetics and Nuclear ChemistryHard
Question
Given the following second order reactions occuring at a certain temperature (T1) :-
(i) A + B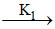 products
products
(ii) C + D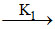 products
products
The activation energy for reaction (i) is Ea1 and for (ii) it is Ea2 such that Ea1 > Ea2 . If the temeprature of both the system is increased from T1 to T2, choose the correct relation from the following :-
(i) A + B
(ii) C + D
The activation energy for reaction (i) is Ea1 and for (ii) it is Ea2 such that Ea1 > Ea2 . If the temeprature of both the system is increased from T1 to T2, choose the correct relation from the following :-
Options
A.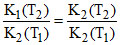
B.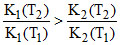
C.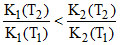
D.None of the above
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Order of reactivity is :-...Which of the following statement is wrong about the rate of reaction?...For a first order reaction A → P, the temperature (T) dependent rate constant (k) was found to follow the equation...For the parallel reactions $A\overset{\quad K_{1}\quad}{\rightarrow}B\text{ and }A\overset{\quad K_{2}\quad}{\rightarrow...Anisotope is :...