Redox and Equivalent ConceptHard
Question
The standard reduction potential for Cu+ / Cu is + 0.34 volt. Calculate the reduction potential at pH = 14 for half cell Cu+2/Cu. Ksp of Cu(OH)2 is 1.0 × 10-19 :-
Options
A.- 0.442 volt
B.- 0.221 volt
C.+0.442 volt
D.None of these
Solution
Ksp = [Cu+2][OH-]2
∴ PH = 14 ⇒ POH = 0
∴ (OH-) = 1 M
∴ 1.0 × 10-19 = [Cu+2] [1]2
[Cu+2] = 10-19 M
Ered = Eored +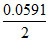 log [Cu+2]
log [Cu+2]
= 0.34 +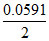 log 10-19
log 10-19
= - 0.221 volt
∴ PH = 14 ⇒ POH = 0
∴ (OH-) = 1 M
∴ 1.0 × 10-19 = [Cu+2] [1]2
[Cu+2] = 10-19 M
Ered = Eored +
= 0.34 +
= - 0.221 volt
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
A mole of N2H4 loses 10 mol of electrons to from a new compound y. Assuming that all the nitrogen appears in the new com...The sum of oxidation states of all carbon atoms in toluene molecule is...The oxidation number of cobalt in K[Co(CO)4] is...Substances which may be oxidized as well as reduced are...In the reaction, Zn + HNO3 → Zn(NO3)2 + NO + H2O, the equivalent weight of HNO3 is...