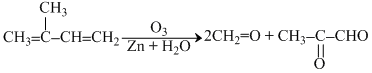Mole ConceptHard
Question
A hydrocarbon C5H8 consumes two moles of hydrogen on catalytic hydrogenation. On ozonolysis, the hydrocarbon produces 2-oxopropanal and methanal (2-moles). The hydrocarbon is-
Options
A.(CH3)2C = C = CH2
B.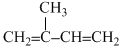
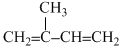
C.CH3-CH2 - C ≡ C - CH3
D.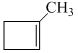

More Mole Concept Questions
Which of the following is not applicable to chemisorption ?...Given the polymers (i) Nylon 6,6 (ii) Buna-S, (iii) Polythene, arrange these in increasing order of their inter-molecula...One mole of the complex compound Co(NH3)5Cl3, gives 3 moles of ions on dissolution in water. One mole of the same comple...Common salt obtained from sea - water contains 96% NaCl by mass. The approximate number of molecules of NaCl present in ...In the reaction2Al(s) + 6HCl(aq) → 2Al3+(aq) + 6Cl-(aq) + 3H2(g) :-...