Basic Maths and Units and DimensionsHard
Question
An ideal gas is initially at temperature T and volume V. Its volume is increased by ᐃV due to an increase in temperature ᐃT, pressure remaining constant. The quantity δ = ᐃV/(VᐃT) varies with temperature as :-
Options
A.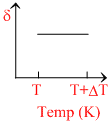

B.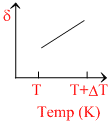

C.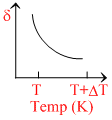

D.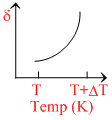

Solution
From ideal gas equation PV = RT .....(i)
or PᐃV = RᐃT
Dividing equation (ii) by (i) we get
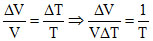 = δ (given)
= δ (given)
∴ δ =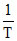 . So the graph between δ and T will be rectangular hyperbola
. So the graph between δ and T will be rectangular hyperbola
or PᐃV = RᐃT
Dividing equation (ii) by (i) we get
∴ δ =
Create a free account to view solution
View Solution FreeTopic: Basic Maths and Units and Dimensions·Practice all Basic Maths and Units and Dimensions questions
More Basic Maths and Units and Dimensions Questions
A body of mass 1 kg begins to move under the action of a time dependent force F→=(2t i^ + 3t2 ...An ideal gas goes through a reversible cycle a → b→ c → d has the V - T diagram shown below. Process d...Two blocks A and B of masses 3 m and m respectively are connected by a massless and 1nextensible string. The whole syste...The x and y coordinates of the particle at any time are x = 5t - 2t2 and y = 10t respectively, where x and y are in mete...A ray of light is incident on an equilateral glass prism (μ = √3) and moves parallel to the base of the prism...