Basic Maths and Units and DimensionsHard
Question
An ideal gas goes through a reversible cycle a → b→ c → d has the V - T diagram shown below. Process d → a and b → c are adiabatic
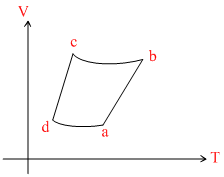

Options
A.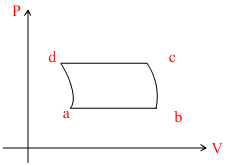

B.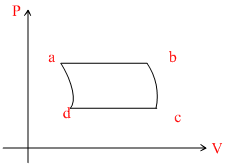

C.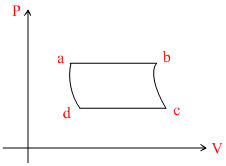

D.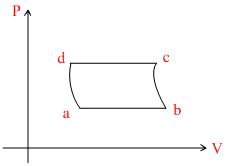

Solution
In VT graph
ab-process : Isobaric line passes through origin, temperature increases.
bc process : Adiabatic, pressure decreases.
cd process : isobaric, volume decreases.
da process : Adiabatic, pressure increase.
ab-process : Isobaric line passes through origin, temperature increases.
bc process : Adiabatic, pressure decreases.
cd process : isobaric, volume decreases.
da process : Adiabatic, pressure increase.
Create a free account to view solution
View Solution FreeTopic: Basic Maths and Units and Dimensions·Practice all Basic Maths and Units and Dimensions questions
More Basic Maths and Units and Dimensions Questions
Three identical bodies are at temperature T1, T2 and T3 having e1, e2 and e3 as their respective emissivities. The therm...A solid sphere rolls down two different inclined planes of same height but of different inclinations, in both cases :-...The matrials sutiable for making electromagnets should have...The ratio of one micron to one nanometre is...Two rods of lengths L1 and L2 are made of materials whose coefficients of linear expansion are α1 and α2. If t...