ThermochemistryHard
Question
Which of the following reaction represent the bond energy :-
Options
A.HCl(g) → 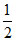 H2(g) +
H2(g) + 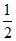 Cl2(g)
Cl2(g)
B.HCl(g) → H+(g) + Cl-(g)
C.HCl(g) → H(g) + Cl(g)
D.2HCl(g) → H2(g) + Cl2(g)
More Thermochemistry Questions
Study the following thermodynamic data given by E. H. P. Cordfunke, A. S. Booji and M. Y. Furkalionk.(i) DyCl3(s) → DyCl...The heat of reaction for : C10H8(s) + 12O2(g) → 10CO2(g) + 4H2O(l) at constant volume is 1228.2 kcal at 25oC. The ...From the following thermochemical equations, find out the bond dissociation enthalpy of CH3−H bond.CH3I(g) → CH3(g) + I(...Calculate the standard free energy change for the ionization HF(aq) → H+ (aq) + F– (aq) from the following data.HF(aq) →...Sublimation energy of Ca is 121 kJ/ mol. Dissociation energy of Cl2 is 242.8 kJ/ mol, the total ionization energy of Ca(...