Nuclear Physics and RadioactivityHard
Question
Mass spectrometric analysis of potassium and argon atoms in a Moon rock sample shows that the ratio of the number of (stable) 40Ar atoms present to the number of (radioactive) 40K atoms is 10.3. Assume that all the argon atoms were produced by the decay of potassium atoms, with a half-life of 1.25 × 109 yr. How old is the rock :-
Options
A.2.95 × 1011 yr
B.2.95 × 109 yr
C.4.37 × 109 yr
D.4.37 × 1011 yr
Solution
19K40 → 20Ar40 + -1e0 + 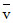
(N0) (0)
(N1) (N2 = N0 - N1)
given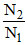 = 10.3 ⇒
= 10.3 ⇒ 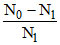 = 10.3
= 10.3
⇒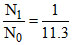 = e-λt
= e-λt
⇒t = ln11.3
⇒t = 4.37 × 109 yrs
(N0) (0)
(N1) (N2 = N0 - N1)
given
⇒
⇒t = ln11.3
⇒t = 4.37 × 109 yrs
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
The activity of a sample reduces from A0 to is one hour. The activity after 3 hours more will :-...The half-life of radioactive element X is the same as the mean lifetime of another radioactive element Y. Initially, bot...The activity of radio active material is :-...The wavelength of Kα X-rays of two metals ′A′ and ′B′ are and respectively, where ′R&...Let NB be the number of β particles emitted by 1 gram of 24 Na radioactive nuclei (half life = 15 hrs) in 7.5 hours...