Nuclear Physics and RadioactivityHard
Question
The wavelength of Kα X-rays of two metals ′A′ and ′B′ are 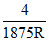 and
and 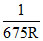 respectively, where ′R′ is Rydberg constant. The number of elements lying between ′A′ and ′B′ according to their aomic number is :
respectively, where ′R′ is Rydberg constant. The number of elements lying between ′A′ and ′B′ according to their aomic number is :
Options
A.3
B.6
C.5
D.4
Solution
For wavelength of Kα n1 = 1 to n2 = 2
and
By solving eq. (i) & (ii) we get
ZA = 26 and ZB = 31
[For elements lie between these two]
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
At some instant two radioactive substance are having amount in ratio of 2 : 1. Their half lives are 12 hrs and 16 hrs th...Photo electric effect is the phenomenon in which...Choose the CORRECT statement(s)...In a hydrogen atom, the electron is in nth exited state. It any com down to second exited state by emitting ten differen...In the hydrogen atom, if the reference level of potential energy is assumed to be zero at the ground state level. Choose...