Work, Power and EnergyHard
Question
The graph which represents the variation of mean kinetic energy of molecules with temperature toC is :-
Options
A.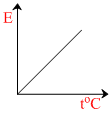

B.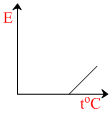

C.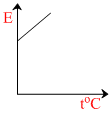

D.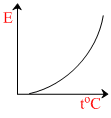

Solution
Mean kinetic energy of gas molecule
E =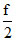 kT =
kT = 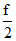 k(t + 273) =
k(t + 273) = 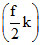 t +
t + 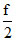 × 273k;
× 273k;
Comparing it with standard equation of straight line y = mx + c. We get m =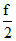 k and c =
k and c = 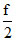 273k
273k
So the graph between E and t will be straight line with positive intercept on E-axis and positive slope with t-axis.
E =
Comparing it with standard equation of straight line y = mx + c. We get m =
So the graph between E and t will be straight line with positive intercept on E-axis and positive slope with t-axis.
Create a free account to view solution
View Solution FreeMore Work, Power and Energy Questions
The velocity of a particle of mass of 1 kg changes from 2 m/s north to 2 m/s east in 2 sec. The change in momentum of th...A bullet fired into a fixed target loses half of its velocity after penetrating 3 cm. How much further it will penetrate...A mass m falls freely from rest. The linear momentum after it has fallen through a height h is (g = acceleration due to ...A block of mass 10 kg is moving in x-direction with a constant speed of 10m/sec it is subjected to a retarding force F= ...A particle of mass m is moving in a circular path of constant radius r such that its centrip-etal acceleration ac is var...