Basic Maths and Units and DimensionsHard
Question
A gas is filled in a cylinder, its temperature is increased by 20% on kelvin scale and volume is reduced by 10%. How much percentage of the gas will leak out at constant pressure :-
Options
A.30%
B.40%
C.15%
D.25%
Solution
Let initial conditions = V,T
and final conditions = V′,T′
By Charle′s law V ∝ T [P remains constant]
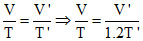 ⇒ V′ = 1.2 V
⇒ V′ = 1.2 V
But as per question, volume is reduced by 10% means V′ = 0.9 V
So percentage of volume leaked out
=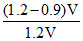 × 100 = 25%
× 100 = 25%
and final conditions = V′,T′
By Charle′s law V ∝ T [P remains constant]
But as per question, volume is reduced by 10% means V′ = 0.9 V
So percentage of volume leaked out
=
Create a free account to view solution
View Solution FreeTopic: Basic Maths and Units and Dimensions·Practice all Basic Maths and Units and Dimensions questions
More Basic Maths and Units and Dimensions Questions
P = θ → Temperature, P → PressureKB → Baltzman constant, Z → DistanceDimension of β is...In the figure ABC is the cross section of a right angled prism and BCDE is the cross section of a glass slab. The value ...Coefficient of linear expansion of brass and steel rods are α1 and α2. Lengths of brass and steel ro...There are two point charges q1 and q2 lying on a circle of unit radius. Electric field intensity at the center of circle...A tuning fork arrangement (pair) produces 4 beats / sec with one fork of frquency 288 cps. A little wax is placed on the...