ThermodynamicsHard
Question
A vessel contains 1 mole of O2 gas (molar mass 32) at a temperature T. The pressure of the gas is p. An identical vessel containing one moles of the gas (molar mass 4) at a temperature 2T has pressure of
Options
A.p/8
B.p
C.2p
D.8p
Solution
pV = nRT or P = 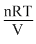 or P ∝ T
or P ∝ T
if V and n are same. Threfore, if T is doubled, pressure also becomes two times i.e., 2 p.
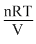 or P ∝ T
or P ∝ T if V and n are same. Threfore, if T is doubled, pressure also becomes two times i.e., 2 p.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
In an isometric change :-...-If DA and BC are adiabatic curves and AB and CD are isothermal curves then : -If DA and BC are adiabatic curves and AB ...In the given figure graph is drawn between and P at different temperature, where n is the numberof moles and R is univer...If one mole of a monoatomic gas (γ = 5 / 3 ) is mixed with one mole of a diatomic gas (γ = 7 / 5 ), the value ...The thermal conductivity of a rod 2.What is its thermal resistivity ?...