Atomic StructureHard
Question
The sodium salt of methyl orange contains 7% of sodium. What is the minimum molecular weight of the compound (atomic weight of Na = 23) :-
Options
A.825
B.359
C.442
D.329
Solution
∵ 7g sodium present in 100 g methyl orange
∴ 23g (one mole atom) sodium present in
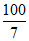 × 23g = 329g methyl organge
× 23g = 329g methyl organge
⇒ molecular weight = 329 amu
∴ 23g (one mole atom) sodium present in
⇒ molecular weight = 329 amu
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Suppose that a hypothetical atom gives a red, green, blue and violet line spectrum. Which jump according to figure would...In [Cu(NH3)4]2+ ion the unpaired electron is present is :-...From the discharge tube experiment, it is concluded that...Photon of which light has maximum energy :...Which of the following has the maximum number of unpaired electrons ?...