Chemical EquilibriumHard
Question
The degree of dissociation of SO3 is α at equilibrium pressure P0.
Kp for 2SO3(g) ⇋ 2SO2(g) + O2(g) is
Kp for 2SO3(g) ⇋ 2SO2(g) + O2(g) is
Options
A.[(P0α3)/2(1 - α)3]
B.[(P0α3)/(2+α)(1 - α)2]
C.[(P0α2)/2(1 - α)2]
D.None of these
Solution
2SO3(g) ⇋ 2SO2 (g) + O2(g)
t = 0 a 0 0
t = teq. a(1-α) aα a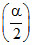
Total mole at eq.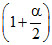
PSO3 =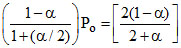 × Po
× Po
PSO2 =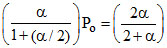 × Po
× Po
PO2 =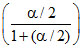 Po
Po
KP =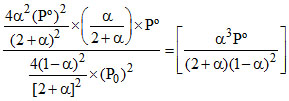
t = 0 a 0 0
t = teq. a(1-α) aα a
Total mole at eq.
PSO3 =
PSO2 =
PO2 =
KP =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
At constant temperature, the equilibrium constant (Kp) for the decomposition reaction N2O4 $\rightleftharpoons$ 2NO2 is...What is the approximate value of log KP for the following reaction?N2(g) + 3H2(g) $\rightleftharpoons$ 2NH3(g) at 25°C. ...The equilibrium constant of the following are : N2 + 3H2 ⇌ 2NH3            K1 N2 + O2 ⇌ 2NO      Â...NH4HS(S) ⇋ NH3(g) + H2S(g)The equilibrium pressure at 25oC is 0.660 atm. What is KP for the reaction....In the system, LaCl3(s) + H2O(g) + heat$\rightleftharpoons$LaClO(s) + 2HCl(g), equilibrium is established. More water va...