KTGHard
Question
The root mean square velocity of the molecules of an ideal gas is :-
Options
A.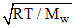
B.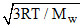
C.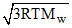
D.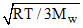
More KTG Questions
At N.T.P. the volume of a gas is changed to one fourth volume, at constant temperature then the new pressure will be : (...Average kinetic energy of a gas molecule depends on-...Consider a collision between an argon molecule and a nitrogen molecule in a mixture of argon and nitrogen kept at room t...A fixed container is fitted with a piston which is attached to a spring of spring constant k. The other end of the sprin...Critical temperature is that temperature...