Hydrogen and its compoundHard
Question
Amongst Ni(CO)4, [Ni(CN)4]2- and NiCl42- are :
Options
A.NiCl4 and NiCl42- are diamagnetic and [Ni(CN) 4]2- is paramagnetic
B.NiCl42- and [Ni(CN) 4]2- are diamagnetic and Ni(CN)4 is paramagnetic
C.Ni(CN)4 and [Ni(CN)4]2- are diamagnetic and NiCl42- is paramagnetic
D.Ni(CN)4 is diamagnetic and NiCN42- and [Ni(CN)4]2- is paramagnetic
Solution
The Valence bond structures of Ni(CO)4, [Ni(Cl)4]2 are as given below :
Ni(CO)4
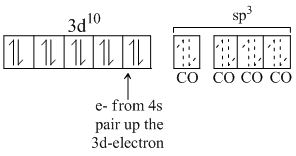
Because there is no unpaired electron, so it will be diamagnetic
[Ni(CN)4]2-
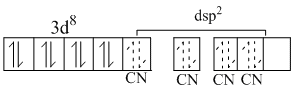
because CN- is strong field ligand so it pairs up the unpaired 3d - electrons and electron oair from CN- enters in 4dsp2h. Os. Because all electrons are paired so it is diamagnetic.
[NiCl4]2-
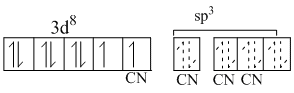
Cl- is weak ligand so it cannot pair up the d-electrons. Due to presence of two unpaired electrons, it is paramagnectic.
Ni(CO)4

Because there is no unpaired electron, so it will be diamagnetic
[Ni(CN)4]2-

because CN- is strong field ligand so it pairs up the unpaired 3d - electrons and electron oair from CN- enters in 4dsp2h. Os. Because all electrons are paired so it is diamagnetic.
[NiCl4]2-

Cl- is weak ligand so it cannot pair up the d-electrons. Due to presence of two unpaired electrons, it is paramagnectic.
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
In nitroprusside ion and NO exist as FeII and NO+ rather than FeIII and NO. These forms can be differentiated by :...Among the following insecticide is :...Anhydrous ferric chloride is preapred by :...Which of the following species participatein sulphonation of benzene ring ?...On heating ammonium dichromate, the gas evolved is :...