Hydrogen and its compoundHard
Question
In nitroprusside ion and NO exist as FeII and NO+ rather than FeIII and NO. These forms can be differentiated by :
Options
A.estimating the concentration of iron
B.measuring the concentration of CN
C.measuring the solid state magnetic moment
D.thermally decomposing the complound
Solution
Presence of Fe as Fe (II) not as Fe (III) and NO as NO+ not as NO, can be confirmed by determining the number of unpaired electrons. If Fe and NO exist as (II) and NO+ then total number of unpaired electrons in the molecule will be 4 (Fe (II) = 4, NO+ = 0). If Fe (III) and NO are present, total number of unpaired electrons will be 6 (Fe (III) = 5, NO = 1). Magnetic moment of a molecule is directly related to the number of unpired electrons and is given by
μ =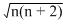 Bohr Magneton
Bohr Magneton
n = No. of unpaired electron
Hwnce, the two cases may be differentiated by measuring magnetic moments.
μ =
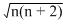 Bohr Magneton
Bohr Magnetonn = No. of unpaired electron
Hwnce, the two cases may be differentiated by measuring magnetic moments.
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
One litre hard water contains 12.00 mg Mg2+, milliequivalents of washing soda required to remove its hardness is. (At. w...Among the following insecticide is :...Which of the following gives propane on hydrolysis?...Which of the vitamins given below is water soluble?...The aqueous solution of the follwoing salts will be coloured in the case fo :...