Solid StateHard
Question
In a body centered cubic cell (bcc) of lattice parameter a, the atomic radius is :-
Options
A.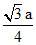
B.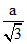
C.a√2
D.a/4
More Solid State Questions
NaCl crystal is...The correct statement(s) regarding defects in solids is(are)...If the edge length of an NaH unit cell is 488 pm. What is the length of an Na-H bond if it crystallises as fcc structure...A molecule A2B (Mw = 166.4) occupies triclinic lattice with a = 5, b = 8, and c = 4 .If the density of AB2 is 5.2 g cm-3...In a solid lattice, the cation has left a lattice site and is located at interstitial position, the lattice defect is :...