p-Block elementsHard
Question
Nitrogen (I) oxide is produced by :
Options
A.thermal decomposition of ammonium nitrate
B.disproportonation of N2O4
C.thermal decomposition of ammonium nitrde
D.interaction of hydroxylamine and nitrous acid
Solution
NH4NO3 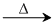 N2O + 2N2O
N2O + 2N2O
NH2OH + (NaNO2 + NCl) → N2O + NaCl + 2N2O
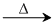 N2O + 2N2O
N2O + 2N2ONH2OH + (NaNO2 + NCl) → N2O + NaCl + 2N2O
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Which of the following hydrides of the oxygen family shows the lowest boiling point?...Aluminium does not react with:...The thermal stability of hydrides of oxygen family is in order :...Which of the following statement is true for sulphur dioxide?...Antiseptics and disinfectants either kill or prevent growth of microganisms. Identify which of the following statement i...