p-Block elementsHard
Question
Which of the following statement is true for sulphur dioxide?
Options
A.It reacts with dry chlorine in absence of moisture to form sulphuryl chloride.
B.It in acidic medium reduces halogens to corresponding halides.
C.Burning magnesium and potassium continue to burn in its atmosphere.
D.All above are correct.
Solution
(A) SO2 + Cl2 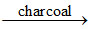 SO2Cl2
SO2Cl2
(B) Cl2 + SO2 + 2H2O → 2HCl + H2SO4
(C) 3Mg + SO2 → 2MgO + MgS ; 4K + 3SO2 → K2SO3 + K2S2O3
(B) Cl2 + SO2 + 2H2O → 2HCl + H2SO4
(C) 3Mg + SO2 → 2MgO + MgS ; 4K + 3SO2 → K2SO3 + K2S2O3
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Which of the following is the increasing order of enthalpy of vapourisation ?...Which of the following element of chalcogen group can form MX2 type of compound whereX = Cl and Br :...H3PO2 $\overset{\quad 140{^\circ}C\quad}{\rightarrow}$ A $\overset{\quad 220{^\circ}C\quad}{\rightarrow}$ B $\overset{\q...Moderate electrical conductivity is shown by :...Select the incorrect statement about the boron....