Chemistry miscellaneousHard
Question
A → product, [A0] = 5M. After 10 minutes reaction is 90% completed. If 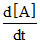 = K[A] then T50% is approximately :-
= K[A] then T50% is approximately :-
Options
A.0.693 min
B.3 min
C.66.0 min
D.0.0693 min
More Chemistry miscellaneous Questions
Which of the following reaction is incorrect :-...The mass of 70% pure H2SO4 required for neutralisation of 500 ml of 2M of NaOH is :-...Reagent used for the above conversion....The structure of XeO3 is :-...For the reaction PCl3 (g) + Cl2 (g) ⇋ PCl5(g), the value of KP at 250oC is 0.61 atm-1. The value of Kc at this tem...