Chemistry miscellaneousHard
Question
The mass of 70% pure H2SO4 required for neutralisation of 500 ml of 2M of NaOH is :-
Options
A.49 g
B.98 g
C.70 g
D.24.5 g
Solution
Meq. of H2SO4 = meq. of NAOH
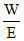 = N × V ⇒ W =
= N × V ⇒ W = 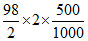 49 g
49 g
But H2SO4 is 70% pure therefore weight
= 49 ×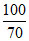 = 70 g
= 70 g
But H2SO4 is 70% pure therefore weight
= 49 ×
Create a free account to view solution
View Solution Free