Periodic TableHard
Question
The correct order of second ionisation potential of carbon, nitrogen, oxygen and fluorine is :
Options
A.C > N > O > F
B.O > N > F > C
C.O > F > N > C
D.F > O > N > C
Solution
On the basis of electronic configuration and effective nuclear charge. The order of I. E2 values may be explained as given below.
E. C. of X(Q, F, N, C), X+ and X2+ are given below:
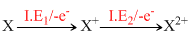
8O : ls2, 2s2, 2p4 ls2, 2s2, 2p3 ls2, 2s2, 2p2
9F : ls2, 2s2, 2p5 ls2, 2s2, 2p4 ls2, 2s2, 2p3
7N : ls2, 2s2, 2p3 ls2, 2s2, 2p2 ls2, 2s2, 2p1
6C : ls2, 2s2, 2p2 ls2, 2s2, 2p1 ls2, 2s2, 2p0
I. E2 value for O is exceptionally higer because E. C. of O+ is half filled and so more stable. Among F+, N+ and C+ removal of electron from F+ requires maximum energy because it has maximum ENC. On the other hand C+ has least ENC, so it needs least energy for the removal of II electron.
E. C. of X(Q, F, N, C), X+ and X2+ are given below:
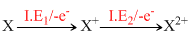
8O : ls2, 2s2, 2p4 ls2, 2s2, 2p3 ls2, 2s2, 2p2
9F : ls2, 2s2, 2p5 ls2, 2s2, 2p4 ls2, 2s2, 2p3
7N : ls2, 2s2, 2p3 ls2, 2s2, 2p2 ls2, 2s2, 2p1
6C : ls2, 2s2, 2p2 ls2, 2s2, 2p1 ls2, 2s2, 2p0
I. E2 value for O is exceptionally higer because E. C. of O+ is half filled and so more stable. Among F+, N+ and C+ removal of electron from F+ requires maximum energy because it has maximum ENC. On the other hand C+ has least ENC, so it needs least energy for the removal of II electron.
Create a free account to view solution
View Solution FreeMore Periodic Table Questions
The hydration energy of Mg2+ is larger than that of :...The formation of the oxide ion O2-(g) requires first an exothermic and then an endothermic step as shown below O(g) + e-...The correct order of $C,N,O$ and F in terms of second ionisation potential is...In which of the following process energy is absorbed:...In period 4 of the periodic table, the elements with highest and lowest atomic radii are respectively....