Atomic StructureHard
Question
The orbital angular momentum of an electron in 2s-orbital is :
Options
A.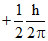
B.zero
C.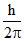
D.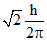
Solution
Orbital angular momentum = 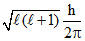 = 0 (since l = 0 for s orbital).
= 0 (since l = 0 for s orbital).
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The configuration [Ar] 3d10 4s2 4p4 is similar to that of...Wave number of the first line in the Balmer series of Be3+ is 2.5 × 105 cm−1. Wave number of the second line of the Pasc...What possibly can be the ratio of the de Broglie wavelengths for two electrons each having zero initial energy and accel...If the radius of first orbit of H–atom is x Å, then the radius of the second orbit of Li2+ ion will be...The difference in angular momentum associated with the electron in two successive orbits of hydrogen atom in :-...