Ionic EquilibriumHard
Question
Solubility product of AgBr at certain temperature is 2.5 × 10-13. Find out solubility of AgBr in grams per litre at this temperature (Ag = 108, Br = 80)
Options
A.2.5 × 10-7
B.5 × 10-7
C.2.5 × 10-13
D.9.4 × 10-5
Solution
Ksp = S2
S =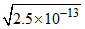 = 5 × 10-7 mole/litre
= 5 × 10-7 mole/litre
AgBr in grams = 5 × 10-7 × 188 = 9.4 × 10-5
S =
AgBr in grams = 5 × 10-7 × 188 = 9.4 × 10-5
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which of the following relations is correct ?...The solubility of sparingly soluble salt A3B2 (molar mass = ‘M’ g/mol) in water is ‘x’ g/L. The ratio of molar concentra...To a 200 ml of 0.1 M weak acid HA solution 90 ml of 0.1 M solution of NaOH be added. Now, what volume of 0.1 M NaOH be a...Buffer capacity of a buffer solution is x, the volume of 1 M NaOH added to 100 mL of this solution if change the pH by 1...The pH of the solution obtanied by mixing 10 mL of 10-1 N HCI and 10 mL of 10-1 N NaOH is:...