Mole ConceptHard
Question
Equal masses of SO2 and O2 are placed in a flask at STP choose the correct statement.
Options
A.The number of molecules of O2 are more than SO2
B.Volume occupied at STP is more for O2 than SO2
C.The ratio of number of atoms of SO2 and O2 is 3 : 4.
D.Moles of SO2 is greater than the moles of O2.
Solution
Let W gas of SO2 and O2 are taken
moles of SO2 =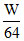
moles of O2 =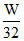
molecules of O2 =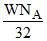
molecules of SO2 =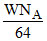
hence molecules of O2 > molecules of SO2
since moles of O2 > moles of SO2, hence volume of O2 at STP > volume of SO2 at STP.
moles of SO2 =
moles of O2 =
molecules of O2 =
molecules of SO2 =
hence molecules of O2 > molecules of SO2
since moles of O2 > moles of SO2, hence volume of O2 at STP > volume of SO2 at STP.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Which of the following statement is correct :...X and Y are two elements which form X2 Y3 and X3Y4. If 0.20 mol of X2Y3 weighs 32.0 g and 0.4 mol of X3Y4 weighs 92.8 g,...Vapour density of a metal chloride is 66. Its oxide contains 53% metal. The atomic weight of the metal is :-...The total number of electrons in one molecule of carbon dioxide is :...If 0.50 mole of BaCl2 is mixed with 0.20 mole of Na3 PO4, the maximum number of moles of Ba3(PO4)2 that can be formed is...