Mole ConceptHard
Question
The correct expression relating molality (m), molarity (M), density of solution (d) and molar mass (M2) of solute is :
Options
A.m = 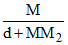 × 1000
× 1000
B.m = 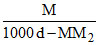 × 1000
× 1000
C.m = 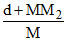 × 1000
× 1000
D.m = 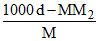 × 1000
× 1000
Solution
Molarity = M
Let volume of be 1 ltr.
∴ mass of solvent = 1000 d - M × M2
Molality = m =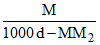 × 1000
× 1000
Let volume of be 1 ltr.
∴ mass of solvent = 1000 d - M × M2
Molality = m =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Vapour density of a metal chloride is 66. Its oxide contains 53% metal. The atomic weight of the metal is :-...A gaseous mixture contains 70% N2 and 30% unknown gas by volume. If the average molecular mass of gaseous mixture is 37....If ‘JAYARAO BANDA’ is written by a graphite pencil, it weighs 3.0 × 10–10 g. How many carbon atoms are present in it? (N...A gaseous alkane is exploded with oxygen. The volume of O2 for complete combustion to the volume of CO2 formed is in 7:4...A hydrocarbon C5H8 consumes two moles of hydrogen on catalytic hydrogenation. On ozonolysis, the hydrocarbon produces 2-...