Mole ConceptHard
Question
112.0 mL of NO2 at STP was liquefied, the density of the liquid being 1.15 g mL-1. Calculate the volume of and the number of molecules in the liquid NO2 . (At. wt. N = 14)
Options
A.0.10 mL and 3.01 × 1022
B.0.20 mL and 3.01 × 1021
C.0.20 mL and 6.02 × 1023
D.0.40 mL and 6.02 × 1021
Solution
Mole of NO2 = 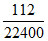 = 5 × 10-3
= 5 × 10-3
Mass of NO2 = 5 × 10-3 × 46 = 0.23 gm
Volume of NO2 =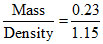 = 0.2 ml
= 0.2 ml
Number of molecule = 5 × 10-3 × 6.023 × 1023 = 3.1 × 1021.
Mass of NO2 = 5 × 10-3 × 46 = 0.23 gm
Volume of NO2 =
Number of molecule = 5 × 10-3 × 6.023 × 1023 = 3.1 × 1021.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
The waste of nuclear power plant contains C12 and C14 in the ratio of 4: 1 by moles. What is the molecular mass of metha...The fractional abundance of Cl35 in a sample of chlorine containing only Cl35 (atomic weight = 34.9) and Cl37 (atomic we...Ethanol is a substance, which is commonly called alcohol. The density of liquid alcohol is 0.8 g/ml at 293 K. If 1.2 mol...The number of F– ions in 4.2 g AlF3 is (Al = 27, F = 19)...On repeated sparking 10 mL of a mixture of carbon monoxide and nitrogen required 7 mL of oxygen for combustion. What was...