ThermochemistryHard
Question
The ᐃHof for CO2(g), CO(g) and H2O(g) are - 393.5 - 110.5 and 241.8 kJ mol -1 respectively. The standard enthaly change (in kJ mol -1 ) for the reaction CO2(g) + H2(g) → CO(g) + H2O(g) is :
Options
A.+524.1
B.+41.2
C.- 262.5
D.- 41.2
Solution
Given that
C + O2 → CO2 ; ᐃHo = - 393.5 KJ ....(i)
C +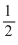 O2 → CO ; ᐃHo = - 110.5KJ ....(ii)
O2 → CO ; ᐃHo = - 110.5KJ ....(ii)
H2 +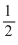 O2 → H2O ; ᐃHo = - 241.8 KJ ....(iii)
O2 → H2O ; ᐃHo = - 241.8 KJ ....(iii)
now making mathematical operation
(ii) + (iii) - (i)
C +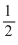 O2 + H2
O2 + H2 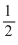 O2 - C - OO2 → CO + HO2O - COO2
O2 - C - OO2 → CO + HO2O - COO2
COO2 + HO2 → CO + HO2O
OR ᐃHo = (-110.5) + (-241.8) -(-393.5)
= + 41.2KJ mol -1
C + O2 → CO2 ; ᐃHo = - 393.5 KJ ....(i)
C +
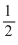 O2 → CO ; ᐃHo = - 110.5KJ ....(ii)
O2 → CO ; ᐃHo = - 110.5KJ ....(ii)H2 +
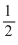 O2 → H2O ; ᐃHo = - 241.8 KJ ....(iii)
O2 → H2O ; ᐃHo = - 241.8 KJ ....(iii) now making mathematical operation
(ii) + (iii) - (i)
C +
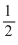 O2 + H2
O2 + H2 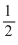 O2 - C - OO2 → CO + HO2O - COO2
O2 - C - OO2 → CO + HO2O - COO2COO2 + HO2 → CO + HO2O
OR ᐃHo = (-110.5) + (-241.8) -(-393.5)
= + 41.2KJ mol -1
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
When 1 g equivalent of strong acid reacts with strong base, the heat released is 13.5 kcal. When 1 g equivalent H2A is c...Which of the following property is correct for the compound having maximum covalent character among the following series...The standard enthalpy of formation of a substance...Heat evolved in the complete combustion of 1.026 kg sucrose at constant pressure will beC12H22O11(s) + 12O2(g) → 12CO2(g...For the reaction C(s) + ½ O2(g) → CO(g),...