ThermochemistryHard
Question
Which of the following statement(s) is/are ture?
Options
A.ᐃE = 0 for combustion of C2H6(g) in a sealed rigid adiabatic container
B.ᐃfHo (S, monoclinic) ≠ 0
C.If dissociation energy of CH4 (g) is 1656 kJ/mol and C2H6(g) is 2812 kJ/mol, then value of C − C bond energy will be 328 kJ/mol
D.If ᐃHf(H2O,g) = - 242 kJ/mol; ᐃHvap (H2O, l) = 44 kJ/mol then ᐃfHo (OH- ,aq.) will be - 142 kJ/mol
Solution
(A) ᐃE will be - ve for combustion reactions
(B) ᐃHfo (S, rhombic) = 0
(C) CH4(g) → C(g) + 4H (g) ᐃH = 1656 kJ/mole = 4 ∈C-H
C2H6(g) → 2C(g) + 6H (g) ᐃH = 2812 kJ/mole = 6 ∈C-H + ∈C-C
So ∈C-C = (2812 -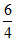 × 1656) kJ/mole = 328 kJ/mole
× 1656) kJ/mole = 328 kJ/mole
(D) Can not be concluded from given data.
(B) ᐃHfo (S, rhombic) = 0
(C) CH4(g) → C(g) + 4H (g) ᐃH = 1656 kJ/mole = 4 ∈C-H
C2H6(g) → 2C(g) + 6H (g) ᐃH = 2812 kJ/mole = 6 ∈C-H + ∈C-C
So ∈C-C = (2812 -
(D) Can not be concluded from given data.
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
When 1 mole of Na(s) is dissolved in large volume of water at 298 K and 1 bar, 184 kJ/mol heat is released. When 1 mole ...Tungsten carbide is very hard and is used to make cutting tools and rock drills. What is the enthalpy of formation (in k...The reaction CH4(g) + Cl2(g) → CH3Cl(g) + HCl (g) has ᐃH = - 25 kcalFrom the given, data, what is the bond e...Calculate the standard free energy of the reaction at 27°C for the combustion of methane using the given dataCH4(g) + 2O...Enthalpy of neutralisation of CH3COOH by NaOH is - 50.6 kJ/mol and the heat of neutralisation of a strong acid with NaOH...