Chemical BondingHard
Question
CO2 is isostructural with :
Options
A.HgCl2
B.C2H2
C.SnCl2
D.NO2
Solution
CO2 is linear with sp-hybridization of C-atom.
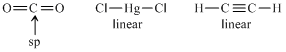
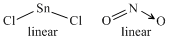
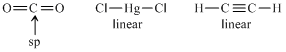
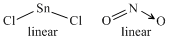
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which of the following are arranged in the decreasing order of dipole moment?...Acidity of diprotic acids in aqueous solutions increases in the order...Which compound exhibits maximum dipole moment among the following ?...Match the following graphs :-GraphA (i) (+ deviation)B (ii) IdealC (iii) (-) deviation...The shape of XeO2F2 molecule is...