Chemical BondingHard
Question
The shape of XeO2F2 molecule is
Options
A.trigonal bipyramidal
B.square planar
C.tetrahedral
D.see-saw
Solution
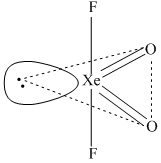
Hybridization = sp3d
Shape = see - saw
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
The maximum radius of sphere that can be fitted in the octahedral hole of cubical closed packing of sphere of radius r i...-Match the following graphs :XA = 1 XB = 1 XB = 0 XA = 0GraphA (i) (+ deviation)B (ii) IdealC (iii) (-) deviation...The tendency of BF3, BCl3 and BBr3 to behave as Lewis acid decreases in the sequence...When two equal sized pieces of the same metal at different temperatures Th (hot piece) and Tc(cold piece) are brought in...The molecule that has linear structure is :...