Chemical BondingHard
Question
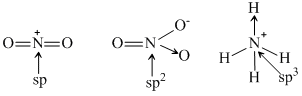
The hybridisation of atomc orbitals of nitrogen in NO2+ NO3- and NH4- are :
Options
A.sp, sp3 and sp2 respectively
B.sp, sp2 and sp3 respectively
C.sp2, sp and sp3 respectively
D.sp2, sp3 and sp respectively
More Chemical Bonding Questions
In which of the following ionization processes, the bond order has increased and the magnetic behaviour has changed?...The molecule having one unpaired electron is :...When two equal sized pieces of the same metal at different temperatures Th (hot piece) and Tc(cold piece) are brought in...Which of the following combination(s) lead(s) to the formation of gerade molecular orbital(s) -...Which of the following species contains equal number of σ - and π- bonds :-...