Chemical BondingHard
Question
Among the following species, identif the isostructural pairs. NF3, NO3-, BF3, H3O+, N3H
Options
A.[NF3, NO3-] and [BF3, H3O+]
B.[NF3, N3H] and [NO3-, BF3]
C.[NF3, N3O+ ] and [NO3-, BF3]
D.[NF3, N3O+ ] and [N3H, BF3]
Solution
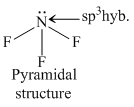
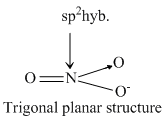
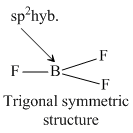
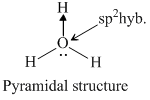
NH3 and H3O+ have sp3 -hybridisation while NO3- and BF3 have sp2 -hybridization. The structure of species given are as follows.KO2- contains O2- ions which contains unpaired electron.
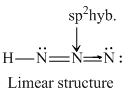




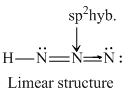
Create a free account to view solution
View Solution Free