ThermodynamicsHard
Question
Benzene burns according to the following equation at 300 K (R = 8.314 J mole-1K-1)
2C6H6(l) + 15 O2 (g) → 12 CO2 (g) + 6H2O(l) ᐃHo = - 6542 kJ/mol
What is the ᐃEo for the combustion of 1.5 mol of benzene
2C6H6(l) + 15 O2 (g) → 12 CO2 (g) + 6H2O(l) ᐃHo = - 6542 kJ/mol
What is the ᐃEo for the combustion of 1.5 mol of benzene
Options
A.-3271 kJ
B.-9813 kJ
C.-4906.5 kJ
D.None of these
Solution
From given reaction ᐃng = 12 - 15 = - 3
so ᐃEo = ᐃHo - ᐃng RT = - 6542 + 3RT
for 1.5 mole, ᐃEo =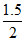 {-6542 + 3RT} = 4912.11 KJ
{-6542 + 3RT} = 4912.11 KJ
so ᐃEo = ᐃHo - ᐃng RT = - 6542 + 3RT
for 1.5 mole, ᐃEo =
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Which of the following processes takes place with decrease of entropy ?...Aluminium is extracted from alumina (Al2 O2) by electrolysis of a molten mixture of...The value of log10K for a reaction A ⇋ B is ( Given : áƒfHo298K = - 54.07 kJ mol-1, áƒrSo298K = 10JK-1mol-1 and R = ...Internal energy does not include:...If an endothermic reaction is non - spantaneous at freezing point of water and becomes feasible at its boiling point, th...