Chemical BondingHard
Question
The molecule which has pyramidal shape is :
Options
A.PCl3
B.SO3
C.CO32-
D.NO3-
Solution
In PCl3, P is sp3 - hybridised with one sp3 h.o. occupied by lone pair.
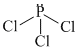
SO3 has planar structure and S-atom is sp2 - hybridised. NO3- and CO32- also have plabar and symmetrical structure with sp2 - hybridised central atom.

SO3 has planar structure and S-atom is sp2 - hybridised. NO3- and CO32- also have plabar and symmetrical structure with sp2 - hybridised central atom.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Molecular AB has a bond length of 1.61(Ao) and a dipole moment of 0.38 D. The fractional charge on eachatom (absolute ma...Which of following is not correctly matched ?...Which one of the following pairs is isostructural (i.e., having the same shape and hyridization) ?...Among the following oxoacids, the correct decreasing order of acid strength is...Match List-I with List-II according to shape.Choose the correct answer from the options given below :...