Chemical EquilibriumHard
Question
When NaNO3 is heated in a closed vessel, oxygen is liberated and NaNO2 is left behind. At equilibrium, NaNO3 (s) ⇋ NaNO2 (s) + 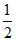 O2 (g)
O2 (g)
Options
A.addition of NaNO2 favours reverse reaction
B.addition of NaNO3 favours forwards reaction
C.increasing temperature favours forward reaction
D.increasing pressure favours reverse reaction
Solution
Addition of solids have no effect on equilibrium and temperature favours endothermic direction while increasing pressure will shift equilibrium in backward direction as ᐃng is +ve.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The equilibrium constant of the following are : N2 + 3H2 ⇌ 2NH3            K1 N2 + O2 ⇌ 2NO      Â...The reaction A(g) + B(g) $\rightleftharpoons$C(g) + D(g) occurs in a single step. The rate constant of forward reaction ...Which one of the following statements is not correct?...For the reaction, 4 NH3(g) + 5 O2(g) ⇋ 4NO(g) + 6H2O(l), ᐃH = positive.At equilibrium which factor will not ...Carbon monoxide in water gas reacts with steam according to the following reaction.CO(g) + H2O(g) $\rightleftharpoons$CO...